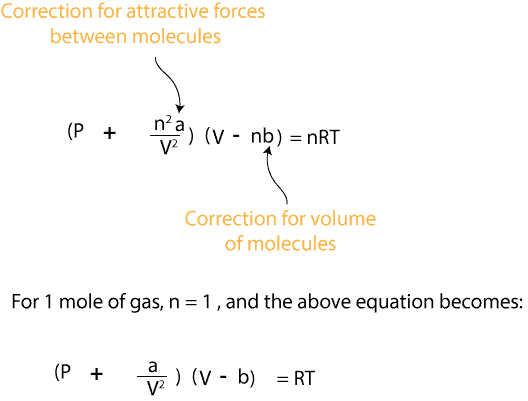homework and exercises - Van der Waals constant $b$ (real gas) chemical form. only - Physics Stack Exchange

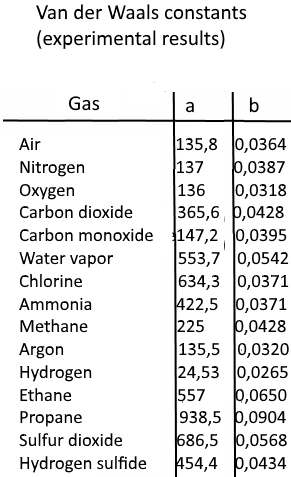
For real gas van der Waals equation is written as: ( p + an^2V^2 ) ( V - nb ) = nRT Where a and b are van der Waals constants.Two sets

Which of the following statements regarding van der Waals' constants a and b is not correct ? (a) The constant a is a measure of van der Waals' forces (b) A gas
Which of the following expressions represent the value and unit of van der Waals' constant a? - Sarthaks eConnect | Largest Online Education Community
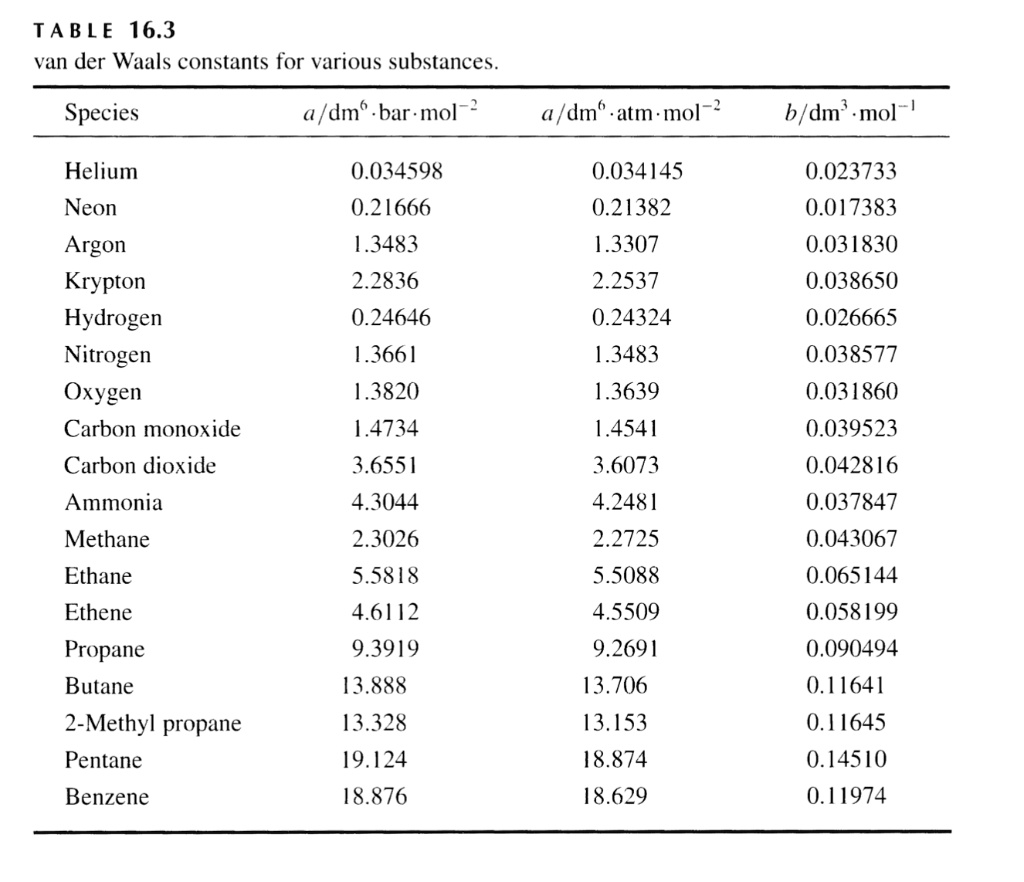
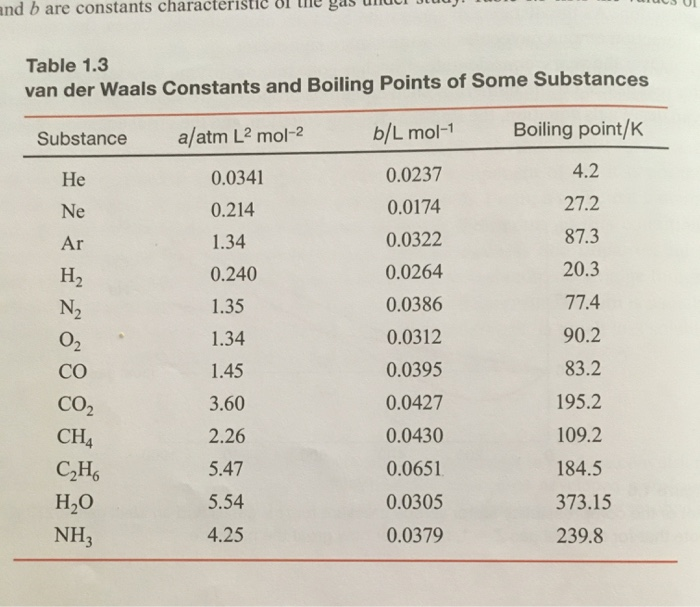
SOLVED:TA BL E 16.3 van der Waals constants for various substances Species /dm6 . bar-mol dm" . atm- mol b/dm'. mol Helium Neon Argon Krypton Hydrogen Nitrogen Oxygen Carbon monoxide Carbon dioxide

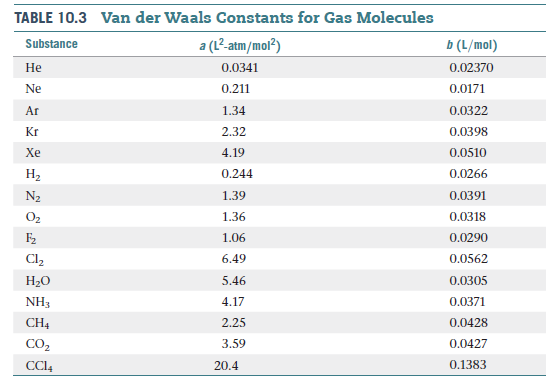
The van der Waals constant for gases A, B, and C are as follows Answer the following: Which gas has the

The values of the van der Waals constants for a gas a = 4.10 dm^(6) "bar" mol^(-2) and b = 0.035 dm^(3) mol^(-

At T=300K, 1.00mol of CO2 occupies a volume of 1.50L. Calculate the pressures given by the ideal gas equation and the van der Waals equation. (The van der Waals constants a and