![IUCr) (E)-4-[(2-Carbamoylhydrazinylidene)methyl]-3-hydroxy-5-hydroxymethyl-2-methylpyridin-1-ium nitrate IUCr) (E)-4-[(2-Carbamoylhydrazinylidene)methyl]-3-hydroxy-5-hydroxymethyl-2-methylpyridin-1-ium nitrate](https://journals.iucr.org/c/issues/2013/07/00/fa3317/fa3317scheme1.gif)
IUCr) (E)-4-[(2-Carbamoylhydrazinylidene)methyl]-3-hydroxy-5-hydroxymethyl-2-methylpyridin-1-ium nitrate

Crystals | Free Full-Text | Bis-Citrullinato Copper(II) Complex: Synthesis, Crystal Structure, and Non-Covalent Interactions

Structural, spectroscopic, and computational characterization of the azide adduct of Fe(III)(2,6-diacetylpyridinebis(semioxamazide)), a functional analogue of iron superoxide dismutase. - Abstract - Europe PMC

Cu2+, Zn2+, and Ni2+ Complexes of C2-Symmetric Pseudopeptides with an Aromatic Central Spacer | Inorganic Chemistry

1H‐1,2,3‐Triazole: From Structure to Function and Catalysis - Lauko - 2017 - Journal of Heterocyclic Chemistry - Wiley Online Library
![Quantification of Nitrate−π Interactions and Selective Transport of Nitrate Using Calix[4]pyrroles with Two Aromatic Walls | Journal of the American Chemical Society Quantification of Nitrate−π Interactions and Selective Transport of Nitrate Using Calix[4]pyrroles with Two Aromatic Walls | Journal of the American Chemical Society](https://pubs.acs.org/cms/10.1021/ja4021793/asset/images/large/ja-2013-021793_0002.jpeg)
Quantification of Nitrate−π Interactions and Selective Transport of Nitrate Using Calix[4]pyrroles with Two Aromatic Walls | Journal of the American Chemical Society

Oxazolidinones and Related Heterocycles as Chiral Auxiliaries/Evans and Post-Evans Auxiliaries | SpringerLink
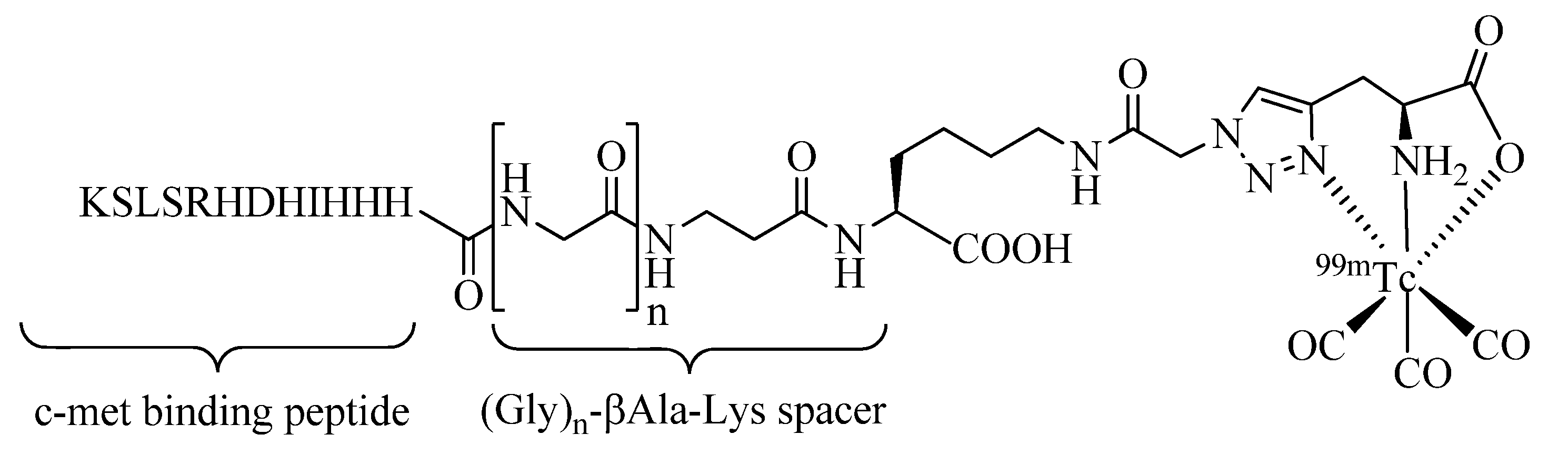
Molecules | Free Full-Text | Click-to-Chelate: Development of Technetium and Rhenium-Tricarbonyl Labeled Radiopharmaceuticals
![Quantification of Nitrate−π Interactions and Selective Transport of Nitrate Using Calix[4]pyrroles with Two Aromatic Walls | Journal of the American Chemical Society Quantification of Nitrate−π Interactions and Selective Transport of Nitrate Using Calix[4]pyrroles with Two Aromatic Walls | Journal of the American Chemical Society](https://pubs.acs.org/cms/10.1021/ja4021793/asset/images/ja4021793.social.jpeg_v03)
Quantification of Nitrate−π Interactions and Selective Transport of Nitrate Using Calix[4]pyrroles with Two Aromatic Walls | Journal of the American Chemical Society

1H‐1,2,3‐Triazole: From Structure to Function and Catalysis - Lauko - 2017 - Journal of Heterocyclic Chemistry - Wiley Online Library

Construction of homo and heteronuclear Ru(II), Ir(III) and Re(I) complexes for target specific cancer therapy - ScienceDirect